Synaptic transmission and its plasticity are not occurring in empty space but in an environment that contains neurons, glial cells and extracellular matrix. Our research primarily focuses on how such interactions of neurons and an abundant subtype of glia (astrocytes) determine hippocampal network function and its dysfunction in disease. The experimental techniques we are mainly using are electrophysiology in its various flavors, two-photon excitation fluorescence microscopy in vitro and in vivo and advanced analyses using custom written software and numerical simulations. See below for examples of typical experiments.
Please also see our recent publications.
Current and past funding: German Research Foundation (DFG, SFB1089, SPP1757 and individual projects), EU (ITN EUGliaPhD), BMBF (SynGluCross), NRW (iBehave, NRW-Rückkehrerprogramm), Human Frontiers Science Programme (HFSP), UCL Excellence Fellowship, DAAD.
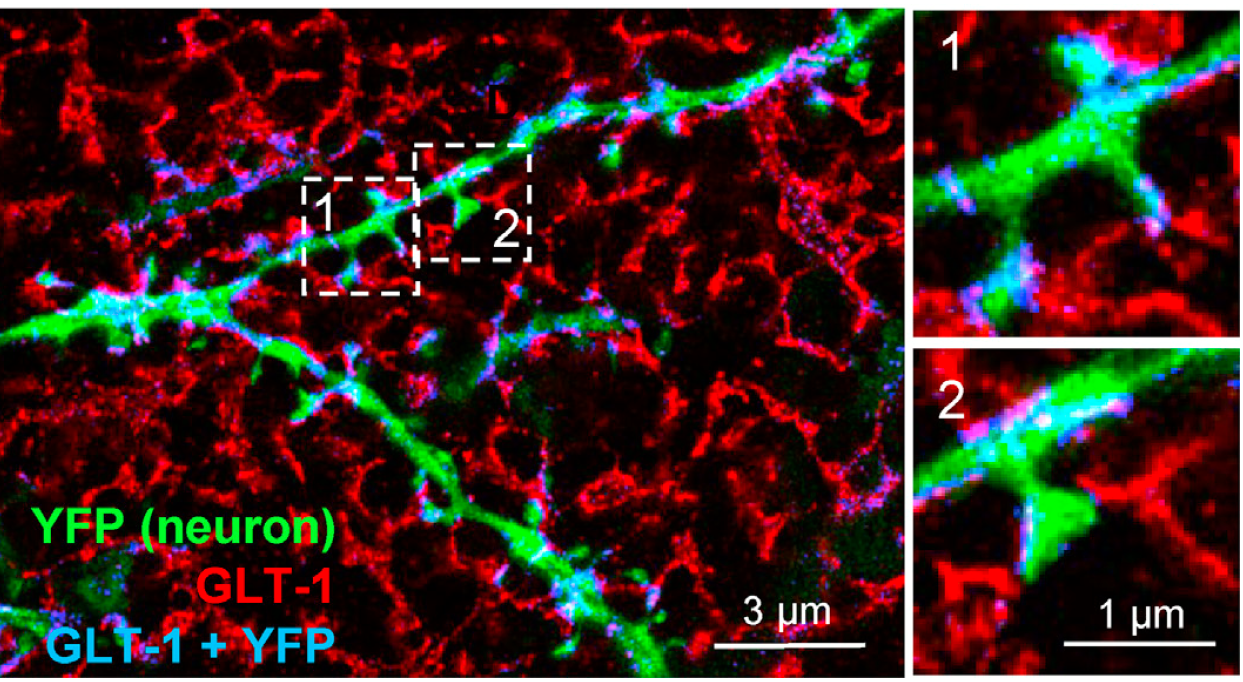 An example of how the fine processes of astrocytes (red) approach synaptic spines on a CA1 pyramidal cell dendrite (green). This was visualized in a recent study using super-resolved expansion microscopy, in which we revealed a principle that governs how strongly astrocytic processes cover individual synapses (also see full paper). Unexpectedly, large and strong synapses were less efficiently covered by astrocytic branches and controlled by glutamate uptake compared to smaller synapses.
An example of how the fine processes of astrocytes (red) approach synaptic spines on a CA1 pyramidal cell dendrite (green). This was visualized in a recent study using super-resolved expansion microscopy, in which we revealed a principle that governs how strongly astrocytic processes cover individual synapses (also see full paper). Unexpectedly, large and strong synapses were less efficiently covered by astrocytic branches and controlled by glutamate uptake compared to smaller synapses.
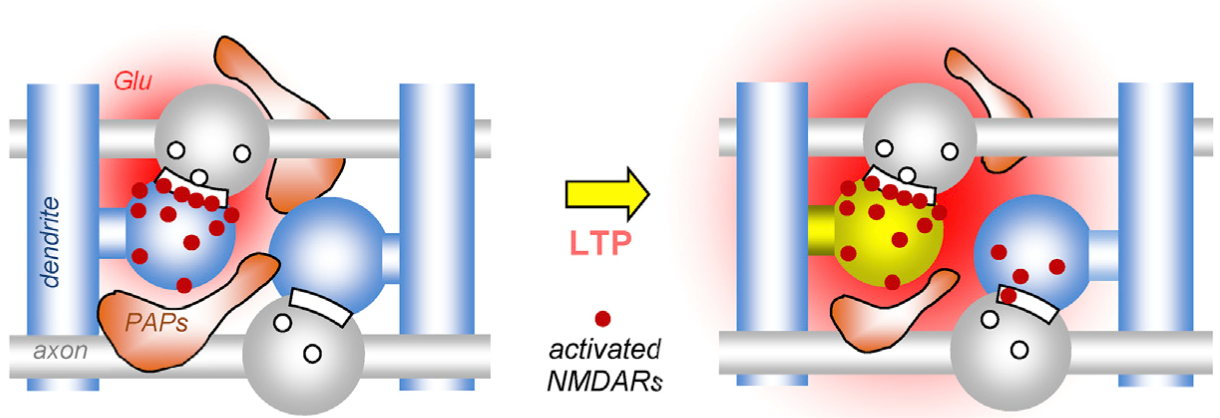 In a related study, we explored which mechanism could lead to this observation. We could uncover that one process that dynamically changes this astrocytic coverage is synaptic plasticity. Interestingly, long-term potentation but not depression induced a rapid withdrawal of perisynaptic astrocyte processes (PAPs, see schematic) from synapses, which reduced the control of glutamate spread in extracellular space and thus the spatial precision of excitatory synaptic transmission (also see full paper).
In a related study, we explored which mechanism could lead to this observation. We could uncover that one process that dynamically changes this astrocytic coverage is synaptic plasticity. Interestingly, long-term potentation but not depression induced a rapid withdrawal of perisynaptic astrocyte processes (PAPs, see schematic) from synapses, which reduced the control of glutamate spread in extracellular space and thus the spatial precision of excitatory synaptic transmission (also see full paper).
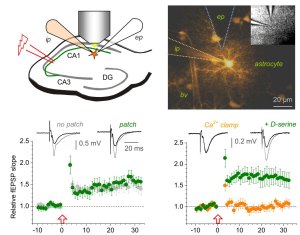 A typical example of an experiment studying how astrocytes support plasticity of synaptic transmission (picture). Synaptic transmission and its plasticity is monitored by stimulating axons while recording field potentials through an extracellular pipette (ep, schematic top left, fEPSP) immediately adjacent to an astrocytes patched in the whole-cell configuration (ip). A sample image (top right) of the astrocyte with its arborization, gap junction coupled neighbors and endfeet outlining a blood vessel (bv). The intracellular signaling of the astrocyte can be manipulated through compounds included in the intracellular solution to test how relevant a particular pathways is, in this case for long-term potentiation (LTP, also see full paper).
A typical example of an experiment studying how astrocytes support plasticity of synaptic transmission (picture). Synaptic transmission and its plasticity is monitored by stimulating axons while recording field potentials through an extracellular pipette (ep, schematic top left, fEPSP) immediately adjacent to an astrocytes patched in the whole-cell configuration (ip). A sample image (top right) of the astrocyte with its arborization, gap junction coupled neighbors and endfeet outlining a blood vessel (bv). The intracellular signaling of the astrocyte can be manipulated through compounds included in the intracellular solution to test how relevant a particular pathways is, in this case for long-term potentiation (LTP, also see full paper).
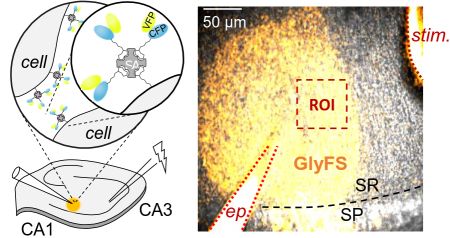 Several current projects investigate the mechanisms that control the extracellular signalling of the NMDA receptor ligands glutamate, glycine and D-serine. In collaboration with Colin Jackson’s lab at the Australian National University we develop and apply novel neurotransmitter sensor. The picture illustrates one approach we have been using successfully. A novel sensor (CFP/YFP) contains a biotin tag that is linked to streptavidin. This mix is then injected into acute hippocampal slices, in which all membranes have been biotinylated previously (left schematic). As a consequence, the sensor is immobilized int the extracellular space and can be visualized using two-photon excitation imaging (right panel, yellow sensor, superimposed on DIC image). We can test under which conditions and neuronal stimulation protocols an NMDAR ligand (here Glycine) is release. See here and here for full papers.
Several current projects investigate the mechanisms that control the extracellular signalling of the NMDA receptor ligands glutamate, glycine and D-serine. In collaboration with Colin Jackson’s lab at the Australian National University we develop and apply novel neurotransmitter sensor. The picture illustrates one approach we have been using successfully. A novel sensor (CFP/YFP) contains a biotin tag that is linked to streptavidin. This mix is then injected into acute hippocampal slices, in which all membranes have been biotinylated previously (left schematic). As a consequence, the sensor is immobilized int the extracellular space and can be visualized using two-photon excitation imaging (right panel, yellow sensor, superimposed on DIC image). We can test under which conditions and neuronal stimulation protocols an NMDAR ligand (here Glycine) is release. See here and here for full papers.
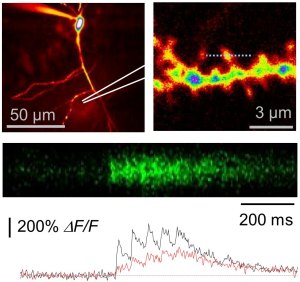 Ca2+ and Na+ signaling can be studied at high temporal and spatial resolution using ion sensitive fluorescent dyes. Shown is an example of Ca2+ imaging in single spines of a CA1 pyramidal cell. Cell are typically filled with a morphological tracer to visualize the morphology (e.g. Alexa Fluor 594). Two-photon excitation microscopy is then used to find spines on oblique dendrites (top left panel). Once a suitable spine is identified a line across that spine is continuously scanned at high frequency (top right panel, 500 Hz). The fluorescence of a Ca2+-sensitive dye (e.g. Fluo-4) is recorded while backpropagating action potentials are elicited by somatic depolarisation. The result are stepwise increases of Ca2+-dependent fluorescence (middle panel, lower panel for analysis). In this particular set of experiments we tested how disrupting the extracellular matrix affects neuronal Ca2+ signaling (also see full paper). We have recently upgraded one setup with a single photon detection system that enables us to measure fluorescence lifetimes (FLIM). Lifetime imaging has several advantages over intensity-based measurements (e.g. insensitivity to bleaching and changing dye concentrations).
Ca2+ and Na+ signaling can be studied at high temporal and spatial resolution using ion sensitive fluorescent dyes. Shown is an example of Ca2+ imaging in single spines of a CA1 pyramidal cell. Cell are typically filled with a morphological tracer to visualize the morphology (e.g. Alexa Fluor 594). Two-photon excitation microscopy is then used to find spines on oblique dendrites (top left panel). Once a suitable spine is identified a line across that spine is continuously scanned at high frequency (top right panel, 500 Hz). The fluorescence of a Ca2+-sensitive dye (e.g. Fluo-4) is recorded while backpropagating action potentials are elicited by somatic depolarisation. The result are stepwise increases of Ca2+-dependent fluorescence (middle panel, lower panel for analysis). In this particular set of experiments we tested how disrupting the extracellular matrix affects neuronal Ca2+ signaling (also see full paper). We have recently upgraded one setup with a single photon detection system that enables us to measure fluorescence lifetimes (FLIM). Lifetime imaging has several advantages over intensity-based measurements (e.g. insensitivity to bleaching and changing dye concentrations).
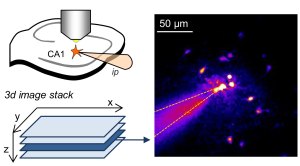 Astrocytes form networks by extensive gap junction coupling that are interwoven with neuronal networks. Diffusible signals like Ca2+ or cAMP and ions can pass between cells from cell to cell in astrocyte networks. This coupling is believed to be variable but at the same time critical for synapse and network function and also relevant for various diseases. The picture shows a sample experiment used to quantify the strength of gap junction coupling using whole-cell patch clamp of astrocytes (top left) and two-photon excitation imaging (right, also see full paper).
Astrocytes form networks by extensive gap junction coupling that are interwoven with neuronal networks. Diffusible signals like Ca2+ or cAMP and ions can pass between cells from cell to cell in astrocyte networks. This coupling is believed to be variable but at the same time critical for synapse and network function and also relevant for various diseases. The picture shows a sample experiment used to quantify the strength of gap junction coupling using whole-cell patch clamp of astrocytes (top left) and two-photon excitation imaging (right, also see full paper).
